Molecular drivers of bladder remodelling in lower urinary tract dysfunction identified through integrated data analysis of patients’ biopsies and animal models
Our Functional Urology Group investigates the
molecular mechanisms underlying the functional and
morphological changes in the bladder during LUTD.
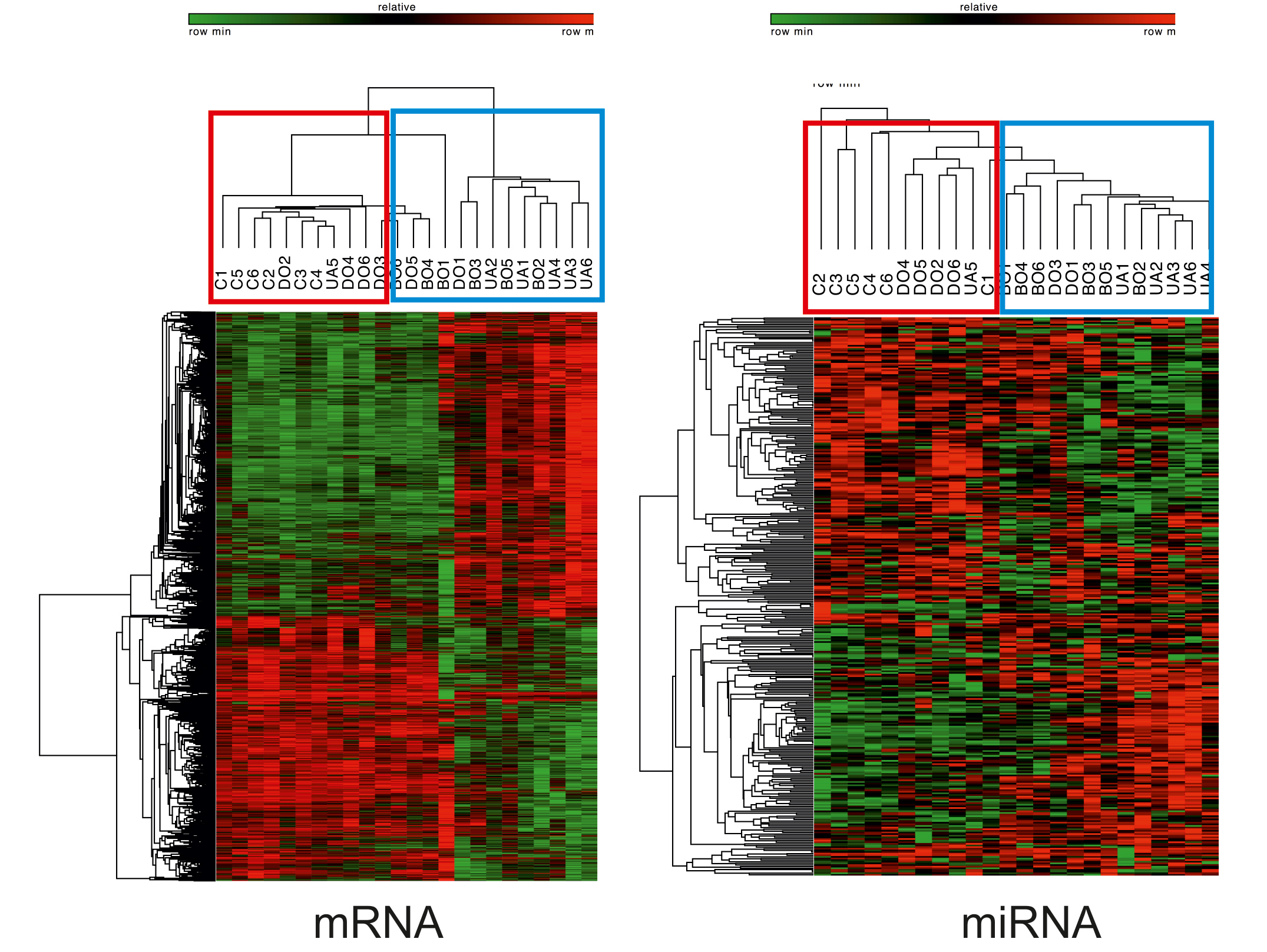
Our comprehensive transcriptome sequencing, the
first of its kind, of human bladder biopsy samples
from patients with benign prostatic obstruction
revealed activation of immune response and
proliferative signalling pathways, and suggested an
increasing involvement of regulatory small
non-coding miRNAs in the control of bladder
function. We identified 3 mRNA- and 3
miRNA-biomarker signatures sufficient to
discriminate between bladder functional states,
validated them in a blinded study and showed the
normalization of their expression in patients whose
bladder function improved after deobstruction. Early
identification of structural changes in the bladder
during LUTD can optimize the timing of treatment. We
are in the possession of a unique collection of
human biopsy samples from patients with
well-characterized bladder functional phenotypes
before and after deobstruction surgery. Many
different underlying pathogenetic mechanisms of
similar symptomatic complexes necessitate different
therapeutic strategies. The functional and molecular
progression of LUTD in the mouse models of pBOO,
SCI, and MS can be monitored longitudinally, in
different phases of the disease characterized by
distinct functional phenotypes. Genes identified in
the animal trials can be compared with the human
biopsy data to further validate the most promising
markers and pathways. Our approach relies on
generation and analysis of big gene expression data
to reveal the triggers of LUTD. To achieve our goal
of unbiased classification of LUTD and
identification of molecular drivers of pathologic
bladder remodelling, we apply machine learning
algorithms to the transcriptome data, clinical
information from the patients, and relevant animal
models.