Targeting PARP-1 Signalling: a novel therapeutic approach for detrusor overactivity
Prerequisite for normal storage and voiding
functions of the urinary bladder is a coordinated
interaction between the muscle components of the
bladder wall (detrusor) and its outlet (urethra and
sphincter). Lower urinary tract dysfunction can be
caused by bladder outlet obstruction and
neurological diseases including spinal cord injury
and multiple sclerosis. Detrusor (bladder muscle)
overactivity (DO) often constitutes a part of an
overactive bladder syndrome, greatly reducing the
patients’ quality of life. High pressure DO after
SCI can irreversibly damage the kidneys. New
therapies preventing the causes of overactive
bladder are urgently needed.
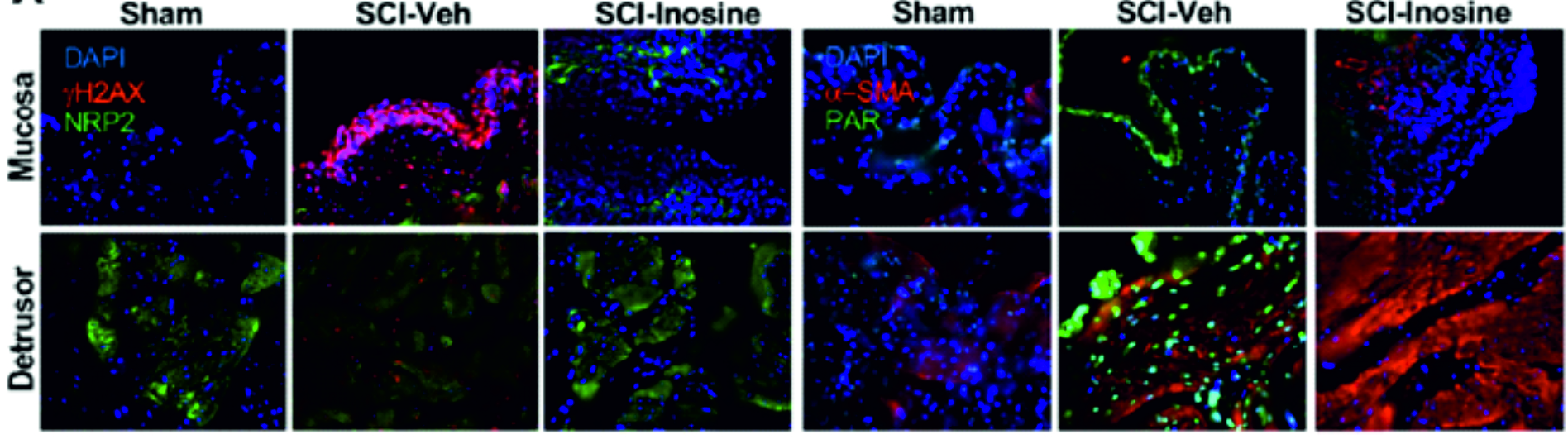
Previously we showed that prolonged treatment
with inosine leads to a significant improvement in
bladder function after SCI. We showed that
poly(ADP-ribose) polymerase 1 (PARP-1) signaling was
increased in the neurogenic bladder and reduced
after treatment with inosine. Here we will
investigate how the activation of PARP-1 signaling
leads to DO, and test the effects of inosine and
pharmaceutic PARP-1 inhibition on the bladder
function. Using the animal models of SCI and MS and
human patients’ material, we will analyze PARP-1
molecular signature in dysfunctional bladders. We
will apply state of the art methods in molecular
biology to discover the activated processes in the
neurogenic bladder and the effects of inhibitors /
drugs. This will allow us to study the cross-talk
between PARP-1 and immune cell activation, and
identify the specific cell types contributing to
detrimental bladder remodeling. Interrupting this
link will allow novel therapeutic approaches, aiming
at removing the cause of DO rather than simply
ameliorating symptoms.