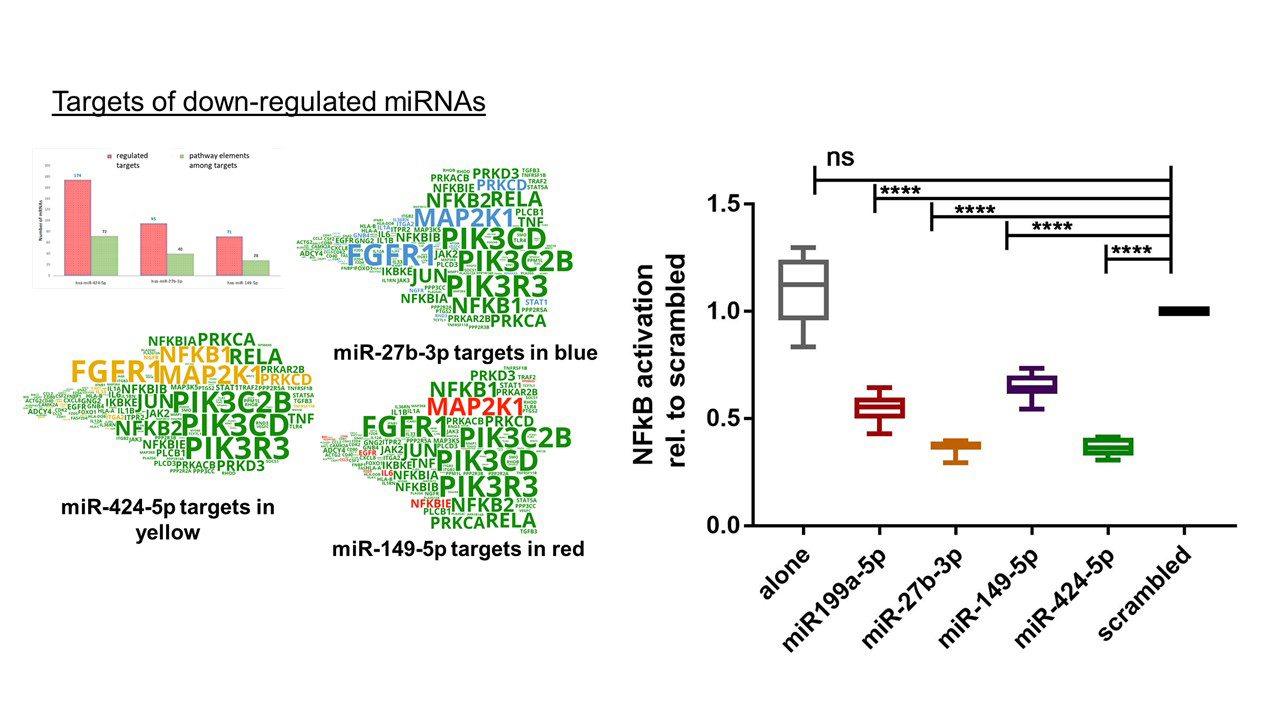
Regulatory potential of miRNAs to mitigate the TNF-alpha-induced bladder dysfunction
Our comprehensive transcriptome analysis showed that TNF-α signalling contributes to the loss of contractility during Benign Prostatic Obstruction (BPO). We hypothesised that a compensatory up-regulation of miRNAs, targeting important hubs of TNF-α signalling pathways might mitigate this process in order to prevent bladder decompensation. We developed and experimentally validated bioinformatic tools to predict the impact of dysregulated miRNAs on cell signalling relevant for disease development. Now we evaluate the significance of the disease-inhibited miRNAs to compensate the aberrant disease-mediated signalling. In this project we address the role of TNF-α in obstructed bladders, focusing on its interplay with miRNAs, which can both regulate gene expression and act as biomarkers of the functional state of the organ. We are investigating the molecular mechanisms of miRNA down-regulation, and are seeking means to preserve the levels of important miRNAs, capable of attenuating pathologic organ remodelling. The potential of down-regulated miRNAs to counteract the aberrant disease-mediated signalling is a highly innovative paradigm. Analysis of miRNA promoter sequences and uncovering the mechanism of their inhibition will contribute to our understanding of bladder hypertrophy and decompensation.